April 2026. Volume 37.
Dermatology Snapshots
Highlights:
A new way of thinking about diagnosing mosaic pigmentary disorders
Nail melanoma and melanonychia - length of monitoring and changes to look out for
Cutaneous immune-related adverse events and oncological outcomes
A Delphi consensus on Topical Steroid Withdrawal
European HS guidelines
Frontal Fibrosing Alopecia in men : a systematic review
Validated tools to help with suspected drug allergies and how to approach allergy labelling
Hydrocolloid dressing vs petroleum ointment application post op - how do they compare?

Beyond the standard: targeted reapplication in palmoplantar scabies of infants
Mussi M et al. Clin Exp Dermatol 2026;51:278–281.
Why we chose this paper?
Scabies treatment failure is increasingly encountered in clinical practice, particularly in infants. This concise report offers a simple, pragmatic modification to standard therapy that is immediately applicable and challenges the assumption that failures are primarily due to resistance.
Study aim and design
A small prospective cohort study (n=22) evaluated whether targeted reapplication of permethrin 5% cream to palms and soles improves outcomes in infants with persistent palmoplantar scabies following standard treatment.
What were the main findings?
All patients had failed standard permethrin regimen (day 0 + day 7 application)
Intervention group (n=11): additional 3 days of targeted palmoplantar application
Control group (n=11): continued standard regimen only
Complete resolution:
100% in intervention group
0% in control group
Behavioural factors (fist clenching, kicking) likely reduced drug contact time
Minimal adverse effects (mild irritation in 2 patients)
Limitations and applicability
Very small sample size with non-randomised allocation
Single-centre study limits generalisability
Short follow-up (4 weeks)
Focused only on infants (<12 months), so applicability to older children/adults is unclear
However, the intervention is low-risk, low-cost, and easily implementable in routine care.
What’s the take-home message?
Standard permethrin regimens may be insufficient in infant palmoplantar scabies.
Targeted reapplication to hands and feet for several consecutive days, based on this small cohort, suggests this can dramatically improve clearance.
Redesign of NHS e-Referral Advice & Guidance: Implications for Dermatology Services
NHS Digital. New Advice and Guidance functionality in the NHS e-Referral Service (2026).
The redesign of Advice and Guidance (A&G) within the NHS e-Referral Service signals a clear shift in how outpatient dermatology services interface with primary care. The updated model moves away from one-off referral queries toward iterative, consultant-led dialogue, supported by structured worklists, templated responses, and improved auditability. In principle, this positions A&G as a front-door triage mechanism, embedding specialist input earlier in the patient pathway.
From a system perspective, the intended benefits are compelling. A&G has the potential to improve referral quality, reduce unnecessary outpatient appointments, and support GP decision-making in real time. For dermatology—where high-volume, low-complexity conditions dominate—this creates an opportunity to standardise advice at scale, particularly for acne, eczema, and lesion assessment. If well implemented, it may also allow more efficient prioritisation of urgent cases and better use of consultant time.
However, commentary in BMJ highlights important concerns that temper this optimism. A key issue is the perception that A&G is being used to suppress referrals, despite assurances that no formal targets exist. There is also a risk of workload redistribution, with primary care managing more patients without proportional resource. For secondary care, increased A&G demand may create a parallel, and potentially unresourced, clinical workload.
More critically, there are clinical governance considerations. Asynchronous advice risks delayed escalation in time-sensitive conditions, including skin cancer, particularly if response times are inconsistent. Additionally, medicolegal responsibility can become blurred between advising consultant and referring clinician.
Overall, the redesign represents a meaningful evolution toward digitally mediated, consultant input at scale. Its success in dermatology will depend less on functionality and more on implementation—specifically, whether A&G is resourced, governed, and used as a clinical tool rather than an administrative filter.

Weekly vs daily bathing for eczema: the Eczema Bathing Study
Bradshaw L et al. Br J Dermatol 2026;194:450–459.
Why we chose this paper?
Bathing advice in eczema is one of the most common—and inconsistently delivered—pieces of guidance in dermatology. Despite its ubiquity, it has been based on weak evidence. This large, pragmatic RCT directly answers a question patients ask daily: how often should I wash?
Study aim and design
This was a UK-based, online, pragmatic randomised controlled trial comparing weekly bathing (1–2 times/week) vs daily bathing (≥6 times/week) over 4 weeks in patients aged ≥1 year with eczema.
n = 438 participants
Primary outcome: eczema symptoms (POEM score)
Secondary outcomes: quality of life, itch, treatment use, global control
Participants were asked not to change other treatments during the study.
What were the main findings?
No clinically meaningful difference in eczema symptoms between weekly and daily bathing
No significant differences in:
Quality of life
Itch severity
Disease control
No safety concerns or increased flares in either group
Results consistent across age groups and baseline severity
Patients can safely choose bathing frequency based on preference
Limitations and applicability
Outcomes were self-reported (POEM) without objective clinician scoring
Short follow-up (4 weeks) limits long-term conclusions
Behaviour and adherence may differ in real-world settings
Conducted in a UK population—however, findings are highly generalisable to NHS practice
Importantly, this was a pragmatic, patient-centred trial, reflecting real-world behaviour rather than idealised conditions.
What’s the take-home message?
Bathing frequency does not meaningfully affect eczema outcomes. Patients can be advised to wash as often as they prefer, simplifying counselling and reducing unnecessary restrictions.
Mobile Dermatoscope Type in Patient-Performed Teledermoscopy: A Study Within a Trial
Ackermann DM et al. JAMA Dermatol. 2026;162(2):124–132.
Why we chose this paper?
Teledermatology and patient-led surveillance are expanding rapidly, particularly in melanoma follow-up. However, there is little practical guidance on which devices patients should use. This study directly addresses a real-world question: does expensive dermoscopy hardware actually add clinical value?
Study aim and design
A randomised study within a trial (embedded in the MEL-SELF trial) compared a low-cost ambient-light dermatoscope with a higher-cost illuminated, polarised dermatoscope for patient-performed teledermoscopy.
n = 251 adults with prior early-stage melanoma
Participants submitted images via smartphone at 3-monthly intervals
Primary outcome: proportion of images adequate for teledermatologist management decisions
What were the main findings?
No significant difference in proportion of patients receiving management recommendations between devices
At baseline:
Polarised: 71.9%
Ambient-light: 67.5%
Over 12 months: similar rates of clinically usable submissions
Image quality slightly better with polarised devices:
Reportable images: 95.0% vs 91.1%
Ambient-light devices had more issues with blur and lighting
Usability similar between groups
Cost difference substantial (~$324 vs ~$35)
Limitations and applicability
Conducted in a motivated melanoma follow-up cohort—may not generalise to broader populations
Participants received training, which may not reflect routine use
Outcome focused on image adequacy, not diagnostic accuracy or clinical outcomes
However, the pragmatic design and real-world teledermatology context enhance relevance.
What’s the take-home message?
Low-cost dermatoscopes are clinically usable for patient-led teledermoscopy.
Higher-cost devices improve image quality—but the marginal gain may not justify the cost in routine practice.

Real-world effectiveness of artificial intelligence–assisted lesion triage on cancer waiting times
Carson L et al. Br J Dermatol. 2026.
Why we chose this paper?
AI triage for suspected skin cancer is being rapidly adopted across the NHS, often with claims of transformative impact. This study is important because it moves beyond accuracy studies and examines real-world system outcomes—specifically cancer waiting times, which are highly relevant to service delivery.
Study aim and design
This was an observational, real-world analysis of NHS England data assessing the impact of the AI triage tool DERM on cancer waiting time performance.
Included 24 NHS trusts using AI triage
Analysed Cancer Waiting Time Faster Diagnosis Standard (28-day target)
Compared pre- and post-implementation performance (2021–2025)
Used regression modelling and meta-analysis to assess changes
What were the main findings?
No overall improvement in waiting time performance across trusts
Substantial variation between sites:
17% showed significant improvement
13% showed significant deterioration
Post-implementation trends:
21% improved over time
17% worsened
Meta-analysis showed no significant pooled effect
High heterogeneity between trusts
Baseline performance did not predict outcomes
Suggests effectiveness is highly context-dependent
Limitations and applicability
Observational design—cannot establish causality
Small number of early-adopter trusts
Potential selection bias (digitally mature sites may differ systematically)
No granular data on workflow integration or patient-level outcomes
However, the use of real NHS performance metrics makes findings highly relevant to UK dermatology services.
What’s the take home message?
AI triage is not a guaranteed solution to dermatology waiting times. Outcomes depend heavily on how and where it is implemented, not just the technology itself.
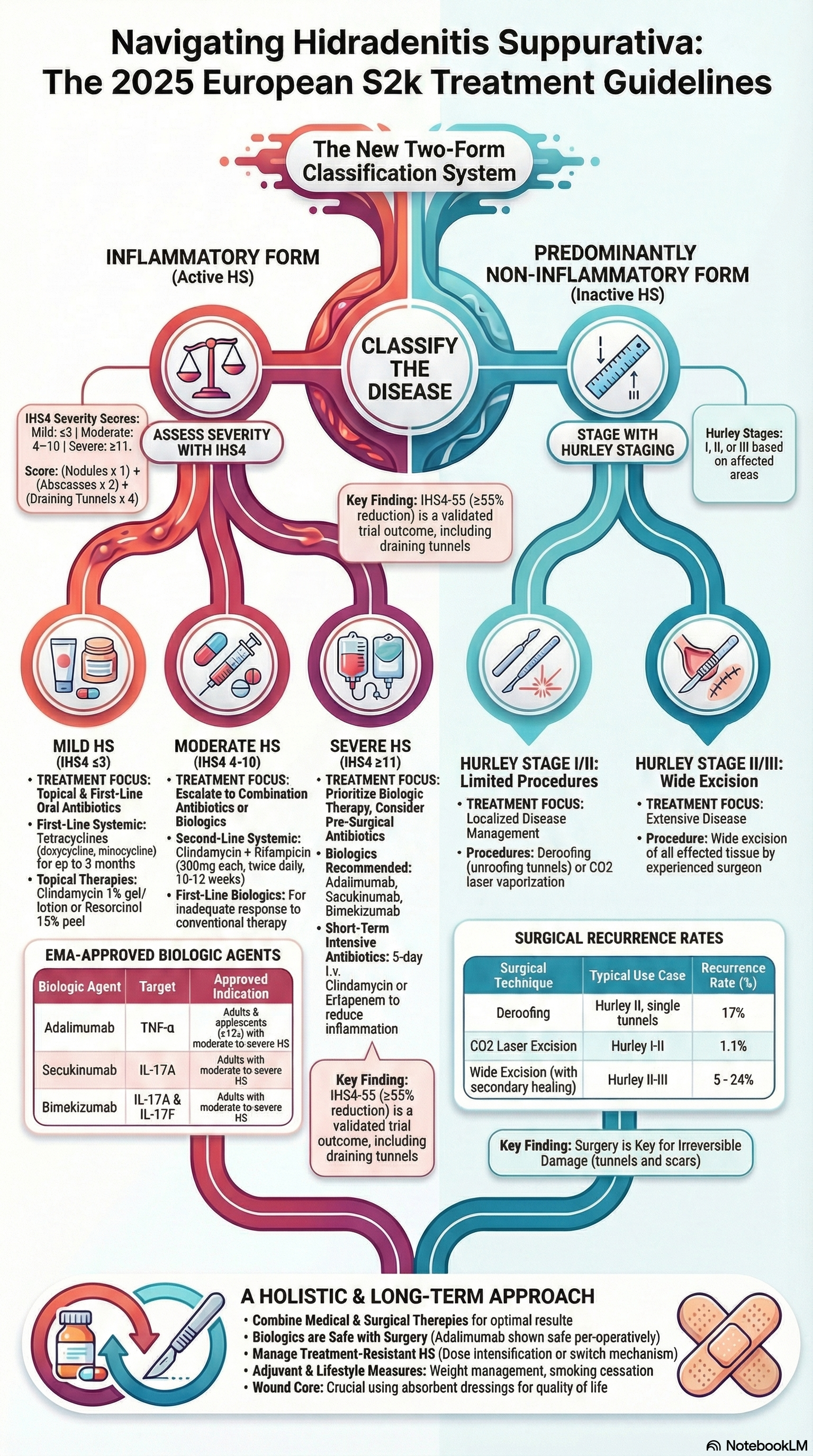
European S2k guidelines for hidradenitis suppurativa/acne inversa – Part 2: Treatment
Zouboulis CC, Bechara FG, Benhadou F, et al. Journal of the European Academy of Dermatology and Venereology. 2025;39:899–941. doi:10.1111/jdv.20472
Background and rationale
Since the last European HS guidelines (S1, 2015), the therapeutic landscape for hidradenitis suppurativa (HS) has changed substantially, driven by validated severity scoring systems and the emergence of effective biologics. This updated S2k guideline (Part 2) focuses specifically on treatment, aiming to provide dermatologists with a practical, evidence-based algorithm that can be applied across disease severities and care settings.
Guideline development and scope
The guideline was developed using a Delphi consensus process among European HS experts and is based on updated literature and clinical trial data. It emphasises stage-adapted therapy, linking treatment choice to validated severity assessment tools. Importantly, HS is reframed into two dominant phenotypes: an inflammatory form and a predominantly non-inflammatory form, which guides medical versus surgical decision-making.
Key therapeutic recommendations
A central update is the endorsement of the IHS4 score to classify inflammatory HS as mild, moderate, or severe, while Hurley staging guides management of non-inflammatory disease. For mild-to-moderate inflammatory HS, oral tetracyclines are highlighted as first-line therapy, with efficacy comparable to clindamycin–rifampicin combinations and a more favourable safety profile. Short-course 5-day intravenous clindamycin is introduced as a strategy to rapidly suppress inflammation and shorten overall antibiotic exposure.
For moderate-to-severe HS, biologic therapy is now firmly embedded in standard care. Adalimumab, secukinumab, and bimekizumab are all EMA-approved options, with clear dosing and response-assessment guidance. Surgery is recommended for irreversible, tunnel-dominant disease and should be combined with anti-inflammatory medical therapy to achieve optimal outcomes.
Clinical relevance
These guidelines provide a clear, practical treatment algorithm grounded in validated scoring systems and real-world trial data. They strongly support early, adequate therapy and combined medical–surgical approaches to reduce disease progression, scarring, and long-term morbidity in HS.

Executive Summary: State-of-the-Art Review: Antibiotic Allergy—A Multidisciplinary Approach to Delabeling.
Mitri EA et al. Clinical Infectious Diseases 2025;81(4):693–694.
Why this paper matters for dermatology
Dermatologists are frequently asked to advise on reported antibiotic allergies, particularly penicillin, in patients with past rashes or unclear drug reactions. This concise review is highly relevant because it highlights validated, easy-to-use clinical risk scores that allow non-allergists to safely assess and often remove incorrect antibiotic allergy labels — improving patient care and antimicrobial stewardship.
What was reviewed
This executive summary synthesises evidence from large cohort studies, clinical trials, and international delabelling programmes. The focus is on practical, bedside approaches to antibiotic allergy assessment that can be used by generalists, hospital physicians, and specialists outside allergy clinics.
Key clinical insights
Over 90% of recorded penicillin allergies are incorrect, yet labels persist for life.
PEN-FAST is a validated, five-item clinical score that reliably identifies low-risk penicillin allergy.
Similar tools now exist for other antibiotics, including CEPH-FAST and SULF-FAST.
Patients classified as low risk can safely undergo direct oral challenge without prior skin testing, with very low rates of adverse reactions.
Modern evidence shows beta-lactam cross-reactivity is mainly due to side-chain similarity, not the beta-lactam ring.
Patients with a history of severe cutaneous adverse reactions (e.g. DRESS, SJS/TEN) remain high risk and should not undergo delabelling outside specialist settings.
Implications for dermatology practice
Dermatologists are well placed to:
Reassure patients with benign childhood or nonspecific rashes
Direct GPs and colleagues to PEN-FAST and similar tools
Help reduce unnecessary avoidance of first-line antibiotics
This is particularly useful when advising on peri-operative antibiotics, infection management, or inpatient consultations.
Bottom line
Validated tools such as PEN-FAST are now widely available and safe to use. Dermatologists should be aware of them and actively signpost colleagues, helping reduce inappropriate antibiotic allergy labels while improving patient outcomes.


